The FFC regime came into force in 2015. Unlike Foods for Specified Health Use (Tokuho), it requires no individual approval — only a notification. The business operator labels the function on its own scientific evidence, at its own responsibility. That makes it usable by SMBs and material houses alike.
But "only a notification" is misleading. The dossier is reviewed in substance; an incomplete file gets rejected, and in the worst case you face a recall recommendation. This article walks through the filing workflow our regulatory team runs day to day.
01The regime end to end
An FFC dossier presents five elements: (1) functional ingredient identification, (2) safety scientific basis, (3) functional scientific basis, (4) manufacturing & QC, (5) label sample. Building the dossier typically runs 6–12 months.
02Material selection — three routes
Three routes to build the functional basis: (A) human trial on the finished product, (B) systematic review of the functional ingredient, (C) using a material with prior filings. For SMBs we recommend (C) → (B). Materials with existing filings dramatically reduce the safety burden.
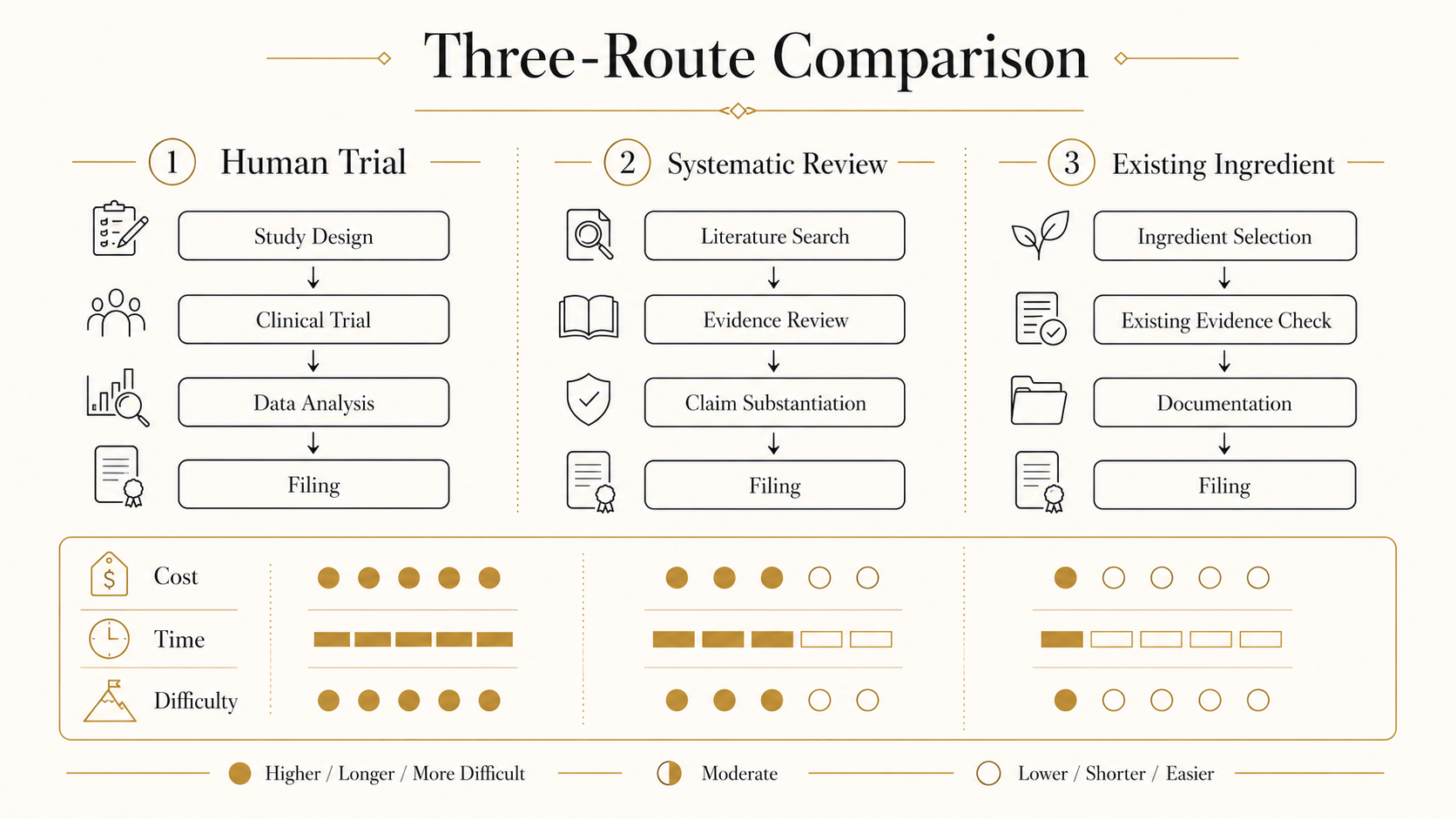
03Systematic review
You need to comprehensively gather human intervention trials from PubMed, Ichushi, JDream III and others, and quality-assess them under PRISMA. Statistical literacy and paper-reading judgement are required; this is usually run with an external partner.
- Search-string design (PICO)
- Database search and de-duplication
- Paper screening (primary/secondary)
- Quality assessment (Cochrane Risk of Bias)
- Qualitative / quantitative synthesis (meta-analysis)
- Review report drafting
Quality of papers matters more than count. A pile of high-bias trials gives you a thin functional basis.
04Safety dossier
Three layers: (1) existing-information assessment, (2) intake-history assessment, (3) safety testing. Long-eaten ingredients can stand on (1) + (2); novel materials need (3) toxicology.
05Dossier structure
Forms I through VII, with attachments: systematic-review report, safety dossier, manufacturing flow, label sample. Total page count: roughly 200–400 pages.

06Submission & publication
File electronically via the CAA notification database. After acceptance, with no rejection within 60 days, the filing is published and you can ship with the claim. On rejection you write a response and re-submit — for a first filing, two or three round trips is normal.
"One-shot submission" is a fantasy. "Plan around two round trips" is the field rule.— MZ Pharma Regulatory
From material selection to filing on your behalf — single point of contact. → Contact us
