OEM (Original Equipment Manufacturer) and ODM (Original Design Manufacturer) sit a single letter apart in English. The scope each side carries, the capability the brand needs to bring, and the total cost shape are completely different. Misframe it before contracting and you end up with "thought it was ODM, turned out to be OEM" or "want OEM but have no formulation."
01Definitions
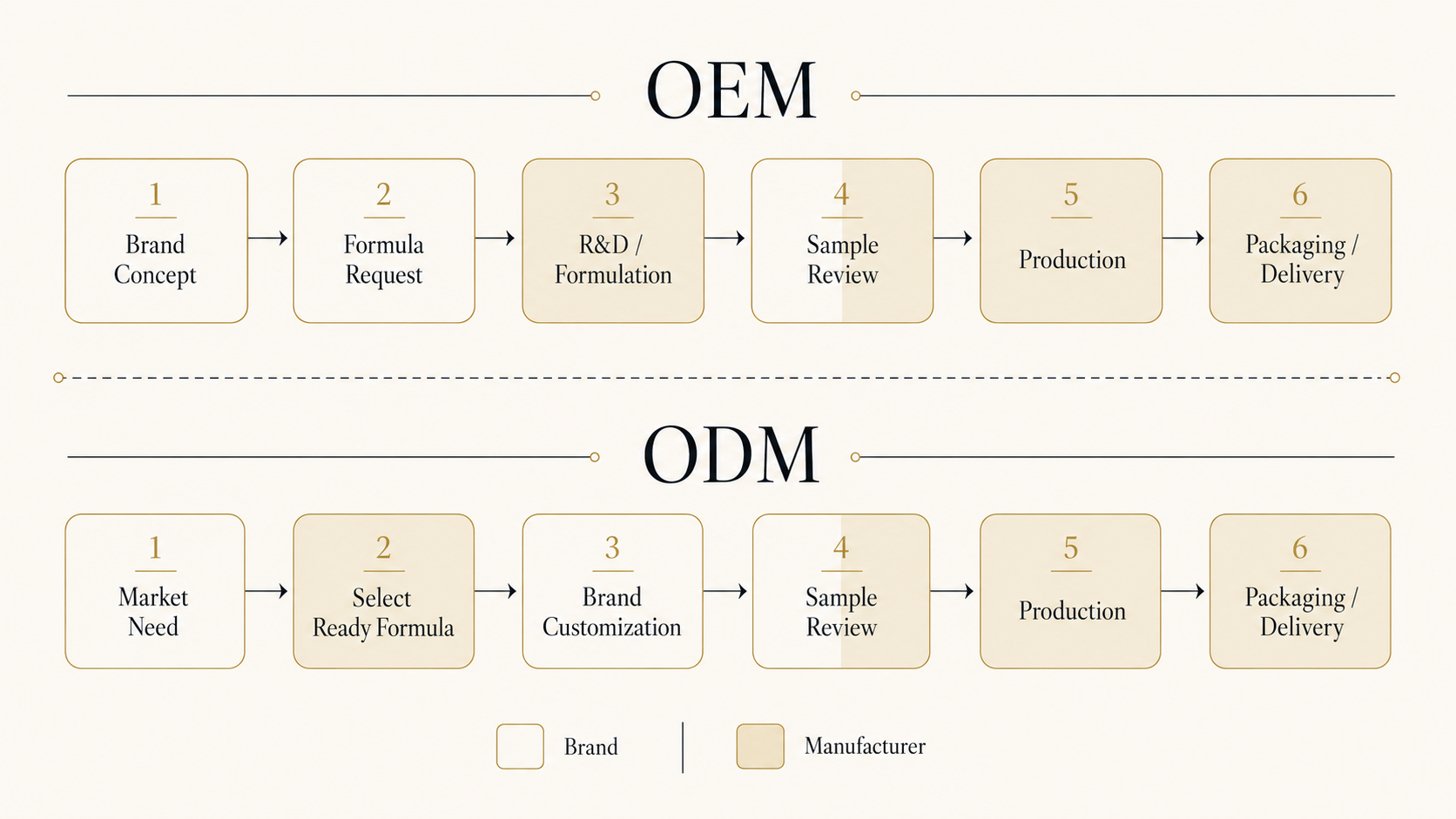
OEM: brand provides formulation/specification, manufacturer produces to spec. Formulation IP belongs to the brand.
ODM: manufacturer provides an existing formulation, brand picks and labels it. Formulation IP sits with (or is shared with) the manufacturer.
| Item | OEM | ODM |
|---|---|---|
| Formulation source | Brand | Manufacturer |
| IP | Brand | Manufacturer |
| Differentiation | High | Mid–Low |
| Initial investment | High | Low |
| Time to market | Slow | Fast |
02Where the responsibility sits
OEM: formulation development, raw-material sourcing, quality-spec decisions → brand carries. Manufacturer's job is to "produce to spec."
ODM: efficacy and safety backing → manufacturer carries (against the existing formulation). Brand owns positioning and marketing.
03Cost structure
OEM: formulation development fee (¥2,000,000+) + prototyping fee (¥500,000+) + production unit cost. Initial investment ¥3,000,000 and up, lower per-unit cost at production.
ODM: formulation usage embedded in unit price. Low initial outlay (sample fee from ¥50,000) but unit cost runs 10–30% higher.
04Speed to market
OEM: formulation through to production typically 9–12 months (longer for kampo lines requiring fermentation).
ODM: formulation pick to production in 3–5 months. Differentiation is capped, but you catch the market window.
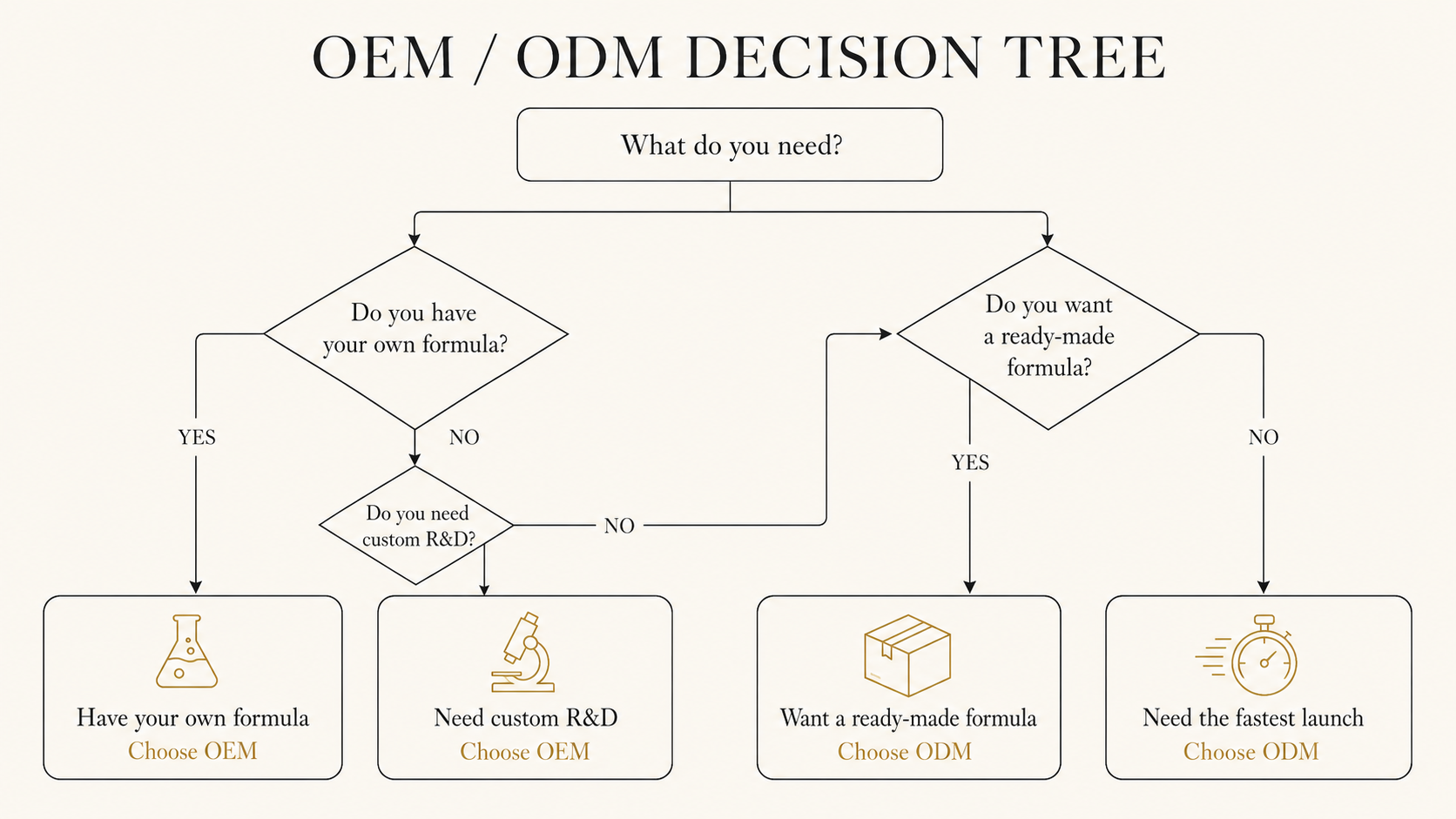
05Decision logic — four scenarios
- Have formulation / capital / want differentiation → OEM
- No formulation / fast launch / cost sensitive → ODM
- Partial formulation / needs reinforcement → hybrid (ODM base + custom ingredient)
- Testing a new category → start ODM, transition to OEM as the brand stabilises
06Why Taiwanese brands pick Made-in-Japan
The Made-in-Japan value: (1) quality halo — Made-in-Japan supports a 20–40% premium; (2) strict GMP/HACCP regime; (3) Foods with Function Claims regime filed in proximity; (4) reverse-export back to Taiwan and Southeast Asia at the premium tier. Costs run higher, but the brand ceiling rises.
For Taiwanese brands, Japanese OEM is an investment, not an expense. We've seen 10-year reverse-export programmes recoup 3–5× revenue.— MZ Pharma OEM Team
From formulation assessment to Japanese CDMO matching — single point of contact. → Contact us
