Microneedles (MN) are devices made of micro-scale needle arrays — 50–1,000 μm. They penetrate the stratum corneum (≈20 μm) but stop short of the pain receptors deeper in the dermis, delivering payloads painlessly. Practical use is spreading: injection replacement, cosmetic enhancement, vaccine delivery.
01Market & growth
Global microneedle-patch market in 2025 is roughly USD 1.5 B, projected to exceed USD 5 B by 2030. In Japan, cosmetics is leading; pharma and wellness are the next waves.
02Four classes of microneedle
By needle structure and mechanism, four families. Use case and drug properties decide which fits.
- Solid — pierces holes in the stratum corneum, then drug is applied for enhanced permeation
- Coated — drug coated on the needle surface, released after insertion
- Dissolving — needle body itself made of sugar or biopolymer, fully dissolves in the skin
- Hollow — needles are hollow and inject liquid (a syringe substitute)
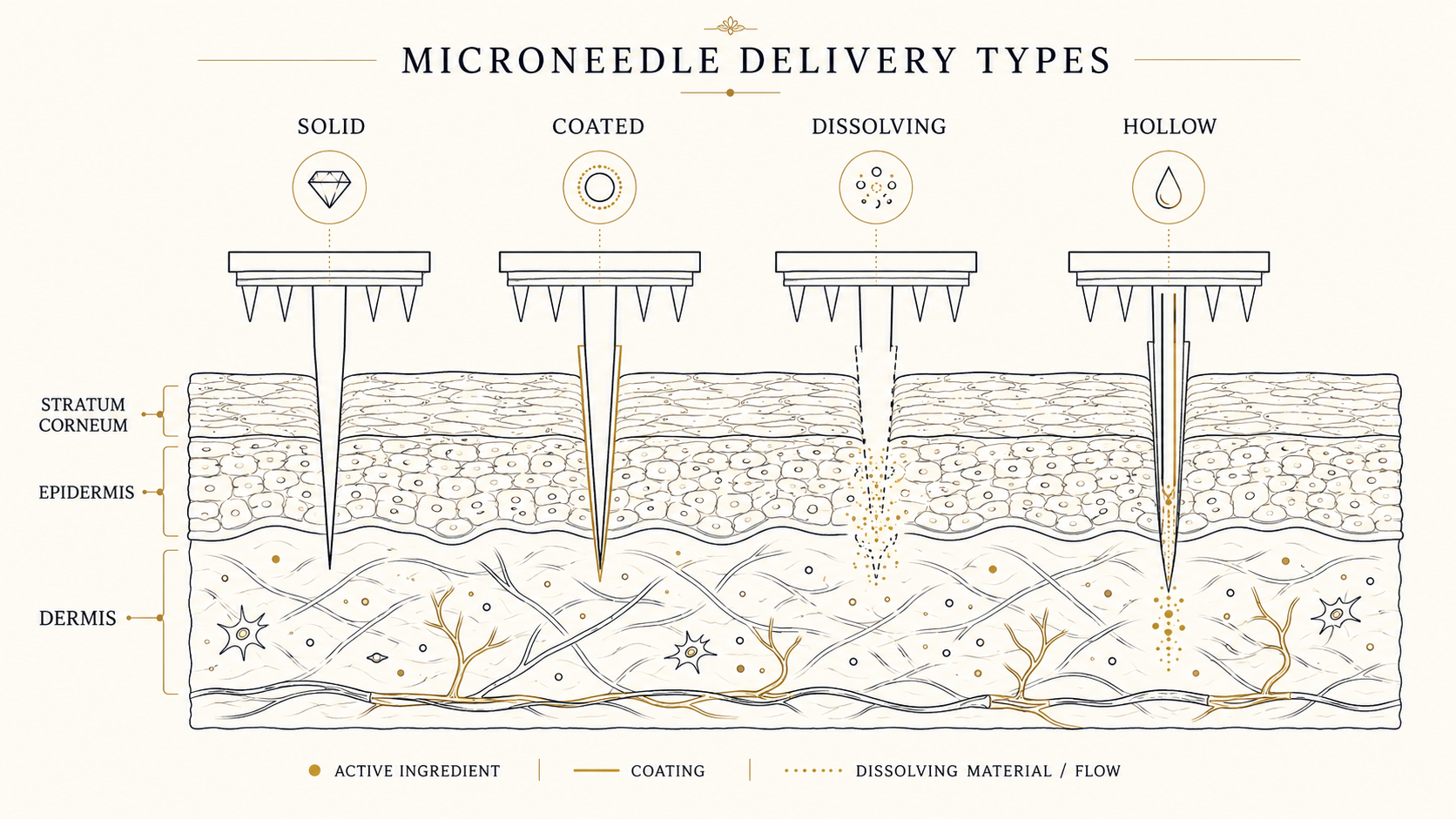
03Materials & needle geometry
Mainstream materials for dissolving microneedles: hyaluronic acid, maltose, chondroitin sulfate and other biocompatible options. Geometries — conical, pyramidal, arrow-tip — affect insertion reliability, retention time and dissolution rate.
Recent two-layer needle structures (active concentrated at the tip) raise drug-utilisation efficiency by 3–5×. Small dose, big effect — a fit for high-value functionals like growth factors and peptides.
04Applications & cases
Four application classes with active commercialisation in 2026:
| Class | Primary actives | Status |
|---|---|---|
| Beauty / skincare | Hyaluronic acid, peptides, vitamin C derivatives | Commercial |
| Pharma (topical) | Anaesthetics, anti-inflammatories, hormones | In approval |
| Vaccines | Influenza, HPV, COVID | Late-stage clinical |
| Functional wellness | Kampo extracts, CBD, plant extracts | Emerging |
05Development hurdles
The technology is maturing but three challenges remain: (1) scaling production, (2) stability and sterility together, (3) ambiguous regulatory classification. "Cosmetic? Quasi-drug? Medical device?" must be settled with a regulatory expert at the product-design stage.
"Inserting a needle into skin" can itself read as a medical device. Needle length and density that ship as a cosmetic have a real practical ceiling.
06Beyond 2026
We see kampo extract × microneedle as the next-generation category, and have ongoing co-development pairing Taiwanese traditional materials with Japanese transdermal technology — kinkotsu-so extract patches for knee, warming kampo blends, eye-area beauty patches, several programmes in parallel.
From "drinking" to "wearing." Re-designing the kampo intake experience accelerates penetration into younger demographics.— MZ Pharma R&D
Concept, formulation, scale-up of functional wellness patches — single point of contact. → Contact us
