"Plant fermentation drink", "kampo enzyme drink", "herbal enzyme" — many names, one common shape: liquid drinks made from many plant raw materials by fermentation and ageing. Japan's market matured through the late 2010s and now competes on the dual axis of health claim and taste experience.
MZ Pharma carries 40 years of accumulated formulation know-how from our Taiwanese parent and runs OEM/ODM through a network of Japanese CDMO partners. This article is the practical decision sheet we hand to brands looking to enter the category.
01The full OEM picture
The flow is six steps: (1) discovery → (2) formulation proposal → (3) prototyping → (4) packaging design → (5) mass production → (6) delivery / QC. Each step has a decision point, and the speed of those decisions sets the overall timeline.
For the first conversation, prepare three things: ① target customer (age, channel), ② positioning axis (beauty / digestion / immunity / sleep), ③ assumed retail price. With those locked in, the first formulation proposal will already land close to reality.
02MOQ & cost structure
Bottom line first. Our standard MOQ is 1,000 bottles at 720 ml-equivalent. That's the first commercial run after prototyping, and the floor for real distribution. For prototype only, 30–100 bottles is workable.
Cost breakdown
Roughly raw materials 35% / fermentation & manufacturing 25% / container 18% / label & carton 10% / testing 5% / logistics 7%. The obvious lever to cut raw-material cost is reducing the number of botanicals — but botanical diversity is precisely what defines an enzyme drink, so cutting there bleeds brand value. Adjust on container, label and logistics first; that's the rule.
Crossing 10k bottles drops the unit container cost by roughly 15–18%. Beyond 30k there's another ~10% gain. Designing the price ladder around "first 1,000 → 10k → 30k" matches a healthy brand-growth curve.
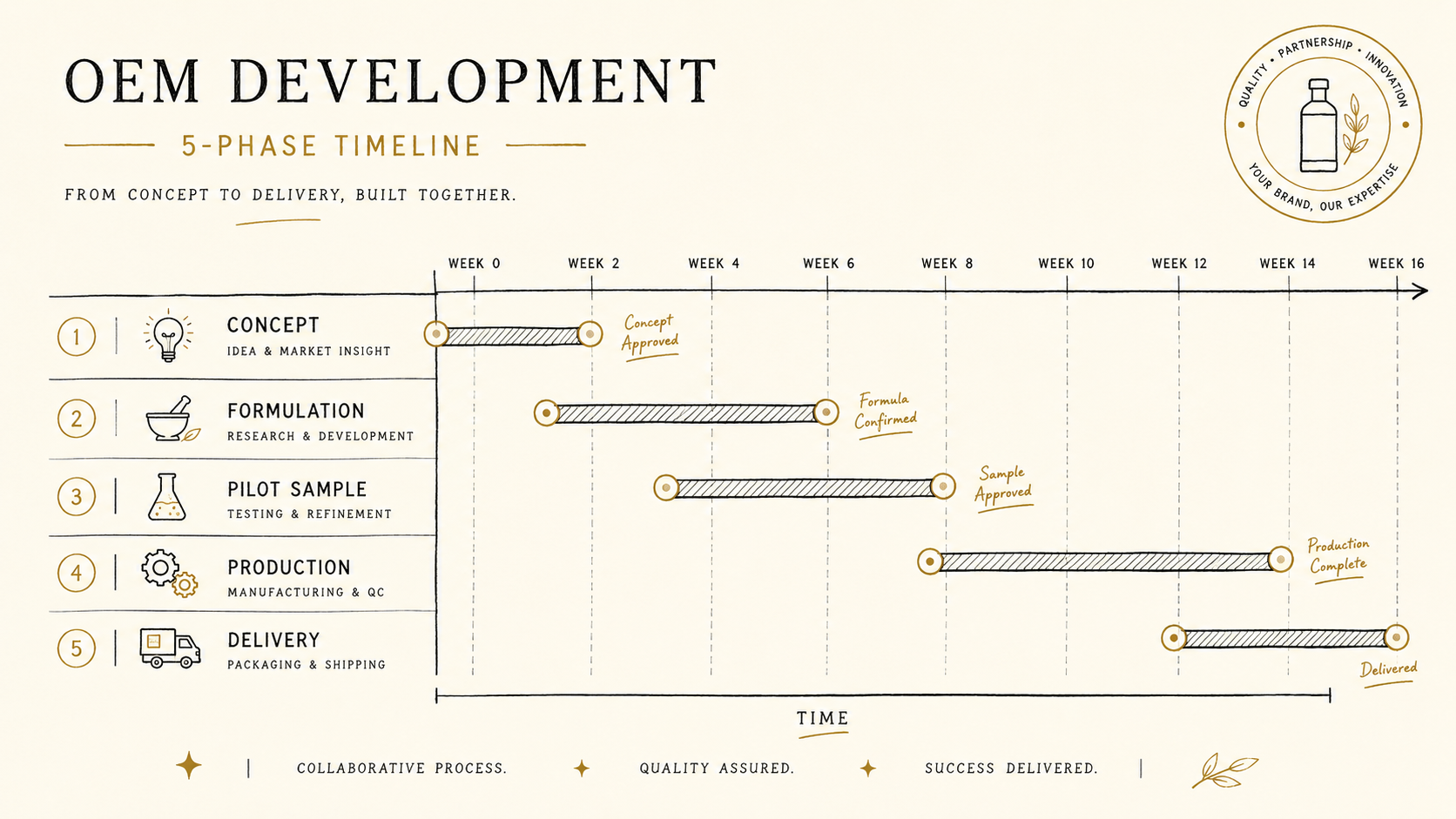
03Timeline: contract to first delivery
Standard lead time is 6–8 months from contract to first delivery. You can compress, but quality and regulatory steps cannot be skipped — the realistic floor is five months.
- Month 1 — discovery, formulation direction agreed, NDA signed
- Month 2–3 — formulation design, prototype round 1 (3–5 samples), sensory evaluation
- Month 3–4 — formulation locked, prototype round 2, container/label design
- Month 4–5 — raw-material procurement, production prep, regulatory final check
- Month 5–6 — production, filling, labelling, testing
- Month 6–8 — delivery, acceptance, channel hand-off
The longest leg is Month 2–3, prototype + sensory evaluation. Fermentation alone takes 2–4 weeks, and running multiple samples in parallel adds time. Not rushing this stage is the single biggest insurance policy against post-launch complaints.

04Formulation & CDMO selection
A formulation has three layers: (A) base fermentate + (B) functional ingredients + (C) flavour adjustment. The base fermentate is the "ground" — dozens of plants, long ageing. Functional ingredients are the layer that carries the positioning axis; in the kampo register, common picks are jujube, goji, reishi, cinnamon bark.
Three criteria for picking a CDMO
- Licences & track record: resident food-sanitation manager, HACCP, annual capacity
- Formulation reproducibility: small sensory drift between prototype and mass run — verify against past cases
- Flexibility: willing to take a 1,000-bottle MOQ, accommodating non-standard containers
We work with multiple CDMOs across Kansai, Kanto and Kyushu and match the partner to production scale and formulation category. "Which house do we use?" is itself a major decision, and that recommendation is the core of our OEM consulting.
05Quality control & regulation
Standard quality control for food OEM runs three checkpoints: incoming-material inspection, in-process inspection, pre-shipment inspection. Tests cover microbiology (total plate count, coliform, mould, yeast), physico-chemistry (pH, Brix, acidity), and visual (sediment, discolouration). For Foods with Function Claims, add quantification of the marker compound.
If you want to claim "Japanese quality", refilling the Taiwan-made fermentate in Japan isn't enough. Fermentation through process control done in Japan is the only story the back label can really carry. — MZ Pharma / Manufacturing
06Common mistakes — and how to avoid them
Mistake 1: prototype OK, mass run tastes different
Almost always the cause is tank size mismatch between prototype and production. A 2 L prototype tank and a 500 L production tank differ in temperature, convection and oxygen profile. Prototype on a small-format machine of the same family as production. That's the rule.
Mistake 2: labels printed, then regulatory says no
Words like "stay beautiful" or "balance from within" can read as drug claims and force a full reprint. Run regulatory review before label sign-off, every time. We bake an internal regulatory check into our label-draft phase.
Mistake 3: ordering exactly the MOQ and stocking out
Launching with 1,000 bottles, hitting harder than expected, stocking out within two months → next run is six months away. It happens. If the channel signal is strong, run +20% on the first batch and pull the next-order lead time into your production plan from day one.
We quote prototype + production from 1,000 bottles. Post-NDA, we send a first proposal: formulation direction × candidate CDMO × indicative timeline. → Contact us
